Why are CHO Cells Critical in Bioprocessing?
"CHO cells" or Chinese Hamster Ovary cells have a long history as tools for scientific discovery. These cells were first introduced to biomedical research in the '50s and currently represent the most frequently used non-human mammalian cell line for the generation of biological therapeutics (i.e., monoclonal antibodies, enzymes, cytokines, and hormones). For example, the majority (~84%) of monoclonal antibody therapeutics approved between 2014 and 2018 were produced using CHO cells (Walsh, 2018). CHO cell-derived monoclonal antibodies have been approved to treat a broad range of indications, including cancer, multiple sclerosis, asthma, HIV, and neuroblastoma.
Other mammalian cell lines typically used in bioprocessing include baby hamster kidney (BHK21) cells and NS0 or Sp2/0 murine myeloma cells (Chin et al. 2019). Nonetheless, several favorable properties have driven CHO cells' increased use in bioprocessing, including their resilience to growth conditions, resistance to viral infections, and high protein synthesis capacity. Significantly, protein-processing by CHO cells more closely conserves the post-translational modifications (e.g., glycosylation) and folding found in human proteins.
Increased use of human cell lines is a more recent bioprocessing trend, including human embryonic kidney 293 (HEK293) cells and HT-1080 human sarcoma cells, among many others (Chin et al. 2019). This approach is increasingly favored due to non-human mammalian cell lines' inability to fully replicate human-like glycosylation patterns. For example, CHO cells do not express several glycosylating enzymes, including GnT-III, Gal alpha2,6 ST, and alpha 1,3/4 fucosyltransferase, present in humans cells (Goh & Ng, 2018). Moreover, mammalian cell lines like CHO cells produce post-translational modifications not present in human proteins, such as the addition of alpha-gal and NGNA glycans. These non-human glycosylations ultimately increase the risk for adverse immunological reactions towards biotherapeutics (Dumont et al. 2016). Additionally, because post-translational modifications influence the yield, activity, and pharmacokinetic properties of recombinant proteins, deficiencies in human-like glycosylation patterns may negatively affect production efforts and therapeutic efficacy.
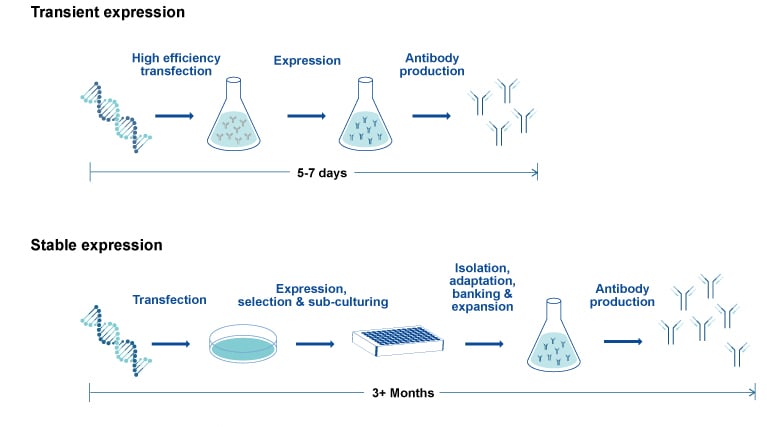
Comparison of workflow and timeline for transient vs. stable antibody expression.
HEK293 vs. CHO Cell Lines in Bioprocessing
HEK293 derived clones are among the most used human cell lines in therapeutic protein production. HEK293 cell clones have been isolated or developed through genetic manipulation and offer improved properties, including increased-transfection efficiency, -growth rate, and -protein production. Perhaps the most significant limitation when using human cell lines is the greater risk for viral contamination and subsequent transmission.
Several CHO cell lines have been developed to date; CHO-K1 and CHOK1SV are frequently used to produce biotherapeutics. CHO cell lines have been genetically engineered to increase productivity, reduce apoptosis, and improve glycosylation (Zhu et al. 2017). While using non-human mammalian cells in bioprocessing carries some disadvantages, when it comes to CHO cells, enough evidence has been accumulated over decades on their safety. CHO cells have been successfully used to produce glycoproteins that are both active and well tolerated by patients (Jayapal et al. 2007). Therefore, CHO cells' predominance in the bioprocessing workflow is projected to continue, backed up by a clear path towards regulatory approvals.
From Transient to Stable CHO Expression
Ultimately, cell line selection for bioprocessing is influenced by factors such as the protein-type being produced and the recombinant protein's glycosylation profile. Significant differences in glycosylation and glycostructure have been reported for proteins produced by CHO vs. HEK293 cells (Croset et al. 2012, Goh & Ng, 2018). Therefore, consistent use of a specific cell expression system across the early and late biotherapeutic protein development stages is critical. For example, the use of CHO transient expression in therapeutic monoclonal antibody development expedites the availability of smaller quantities of antibody candidates, ideal for preliminary in vitrotesting. Higher-yield stable CHO expression of lead recombinant monoclonal antibodies is a recommended path towards later preclinical stages. This approach increases the probability that candidates for pharmacological and toxicological characterization conserve established glycosylation patterns supporting IND and therapeutic development (Jain et al. 2017, Sifiniotis et al. 2019).
Reference
Chin, C. L. et al. A human expression system based on HEK293 for the stable production of recombinant erythropoietin. Sci. Rep. (2019) doi:10.1038/s41598-019-53391-z.
Croset, A. et al. Differences in the glycosylation of recombinant proteins expressed in HEK and CHO cells. J. Biotechnol. (2012) doi:10.1016/j.jbiotec.2012.06.038.
Dumont, J., Euwart, D., Mei, B., Estes, S. & Kshirsagar, R. Human cell lines for biopharmaceutical manufacturing: history, status, and future perspectives. Critical Reviews in Biotechnology (2016) doi:10.3109/07388551.2015.1084266.
Goh, J. B. & Ng, S. K. Impact of host cell line choice on glycan profile. Critical Reviews in Biotechnology (2018) doi:10.1080/07388551.2017.1416577.
Jain, N. K. et al. A high density CHO-S transient transfection system: Comparison of ExpiCHO and Expi293. Protein Expr. Purif. (2017) doi:10.1016/j.pep.2017.03.018.
Jayapal, K. P., Wlaschin, K. F., Hu, W. S. & Yap, M. G. S. Recombinant protein therapeutics from CHO Cells - 20 years and counting. Chem. Eng. Prog. (2007).
Sifniotis, V., Cruz, E., Eroglu, B. & Kayser, V. Current Advancements in Addressing Key Challenges of Therapeutic Antibody Design, Manufacture, and Formulation. Antibodies (2019) doi:10.3390/antib8020036.
Walsh, G. Biopharmaceutical benchmarks 2018. Nat. Biotechnol. (2018) doi:10.1038/nbt.4305.
Zhu, M. M., Mollet, M., Hubert, R. S., Kyung, Y. S. & Zhang, G. G. Industrial Production of Therapeutic Proteins: Cell Lines, Cell Culture, and Purification. in Handbook of Industrial Chemistry and Biotechnology (2017). doi:10.1007/978-3-319-52287-6_29
- Like (2)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



