Tapping the Enhanced Potency of “Hyper-IL-6” to Regenerate Spinal Neurons after Injury
Spinal cord injuries affect ~17,810 individuals each year in the USA, with no cure yet in sight. Based on the National Spinal Cord Injury Statistical Center, spinal cord injuries often result from vehicular accidents and falls, accounting for 38.6% and 32.2% of injuries since 2015. Currently, in the US, over 170 spinal injury-related clinical studies are actively recruiting for a broad range of interventions, including surgical procedures, cell therapies, devices, drugs, and behavioral therapies (ClinicalTrials.gov). Some of these studies aim to restore motor function. Other studies seek to solve or reduce a wide variety of spinal cord injury complications such as inflammation, neuropathic pain, muscle atrophy, and neurogenic bladder.
Aftermath of Spinal Cord Injury
The cellular and molecular pathophysiological changes following spinal cord injuries depend on the type and severity of the trauma. More frequently, accidental trauma results in backbone fracture or crushing, leading to compression and stretching of the spinal cord and blood vessels. These traumatic events alter the spinal cord’s integrity, damaging motor axons, oligodendrocytes, and blood vessels. Subsequently, a state of increased excitability and inflammation leads to prolonged damage by inducing cell death of neurons, oligodendrocytes, and astrocytes. Ultimately, glial activation gives rise to scaring around the lesion’s cavity, which plays a role in reducing further damage but also inhibits axonal regrowth or regeneration (Tran et al. 2017, Katoh et al. 2019).
Restoring Motor Function
Much effort has been devoted to understanding the cellular and molecular mechanisms controlling axonal regrowth. However, a critical obstacle to all strategies aiming at restoring motor function is the low capacity of neurons in the adult central nervous systems (CNS) to regenerate axons following injury.
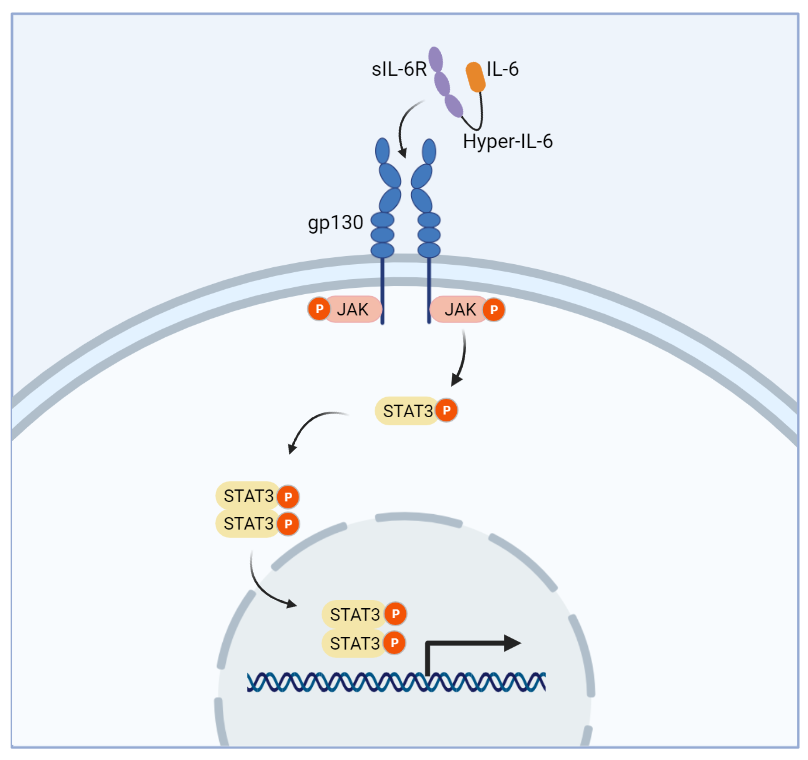
Hyper-IL-6 is a designer cytokine developed from the fusion of IL-6 and the soluble IL-6 receptor alpha subunit by a peptide linker. Hyper-IL-6 binds and potently activates the gp130 receptor inducing JAK/STAT3 signaling and neuronal regeneration in the spinal cord.
Recent work by a team at Ruhr University of Bochum, Germany, expressing a modified form of IL-6 in the sensorimotor cortex, shows promising results inducing regeneration and restoring function in a mouse model of spinal cord injury. Briefly, after generating a severe spinal cord injury, Leibinger et al. induced cortical expression of a modified form of IL-6, a fusion of IL-6 and its soluble receptor alpha subunit (IL-6/sIL-6R). The IL-6/sIL-6R fusion or “hyper-IL-6” was shown previously to activate gp130 mediated signal transduction effectively and induce regeneration in an optic nerve injury model (Peters et al. 1998, Leibinger et al. 2016).
Leibinger et al. expressed the highly potent hyper-IL-6 to overcome the limited expression of the IL-6 alpha receptor subunits in the CNS, inducing binding to gp130 and activation of the JAK/STAT3 regenerative pathway. They found that transduction of the sensorimotor cortex with AAV1-hyper-IL-6 effectively induced STAT3 activation in cortical tissue. Additionally, hyper-IL-6 induced significant corticospinal tract regeneration and supported hindlimb motor function recovery by eight weeks following spinal injury. Most importantly, Leibinger and colleagues found that transneuronal hyper-IL-6 promotes regeneration of serotonergic fibers passed lesion sites and that this effect may indeed underscore the observed functional recovery.
Given the complexities of motor control connections, much remains to be elucidated about the mechanisms underscoring hyper-IL-6 induced regeneration and functional recovery. However, these findings support that hyper-IL-6 serves as a potent factor that may be combined with other strategies to improve overall regenerative and functional outcomes following spinal cord injury.
Reference
Katoh, H., Yokota, K. & Fehlings, M. G. Regeneration of spinal cord connectivity through stem cell transplantation and biomaterial scaffolds. Frontiers in Cellular Neuroscience (2019) doi:10.3389/fncel.2019.00248.
Leibinger, M. et al. Boosting central nervous system axon regeneration by circumventing limitations of natural cytokine signaling. Mol. Ther. (2016) doi:10.1038/mt.2016.102.
Leibinger, M. et al. Transneuronal delivery of hyper-interleukin-6 enables functional recovery after severe spinal cord injury in mice. Nat. Commun. (2021) doi:10.1038/s41467-020-20112-4.
Peters, M. et al. In vivo and in vitro activities of the gp130-stimulating designer cytokine Hyper-IL-6. J. Immunol. (1998). PMID: 9759879
Tran, A. P., Warren, P. M. & Silver, J. The biology of regeneration failure and success after spinal cord injury. Physiological Reviews (2018) doi:10.1152/physrev.00017.2017.
- Like (2)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



