Severe Respiratory Infections: New Mechanism for Pathology
Severe respiratory illnesses, such as those resulting from influenza virus infections, are often associated with significant mortality. The CDC has estimated the annual burden in the US to be around 12,000-61,000 influenza-related deaths since 2010. Understanding the molecular mechanisms underscoring severe respiratory illness, such as acute respiratory distress syndrome (ARDS), will facilitate the development of effective therapeutic strategies to curve lung tissue damage.
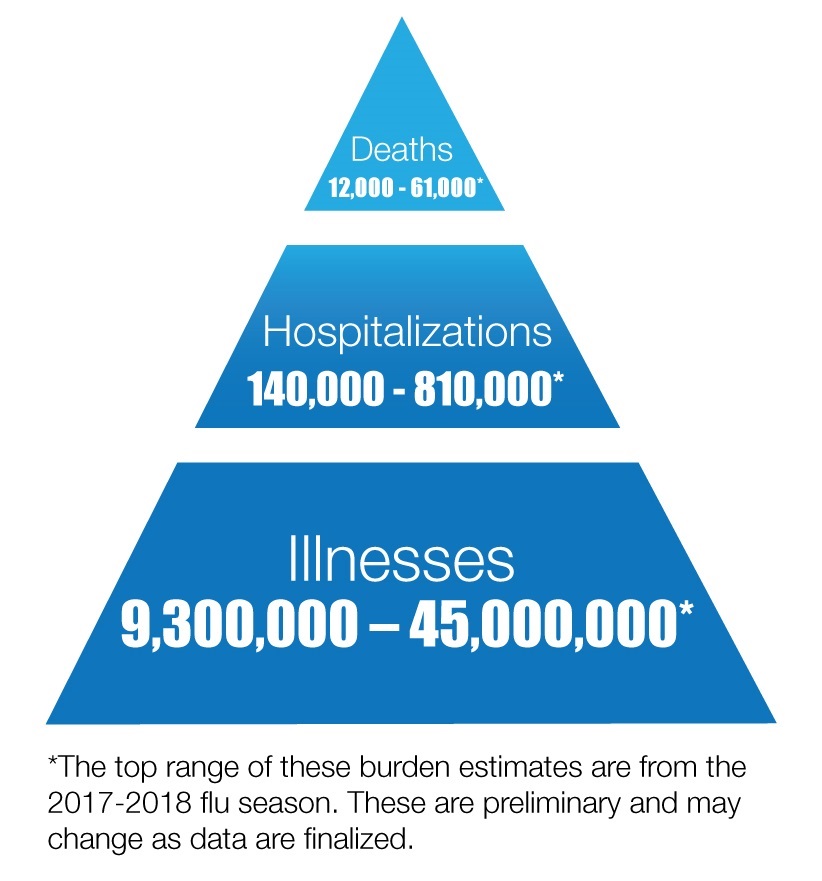
"Source: CDC"; "Materials developed by CDC."
Recent work by a group at New York University and the University of Alberta has provided new insights into the molecular mechanisms leading to inflammation and lung tissue damage following influenza infections.
High-Mannose as a Marker of Influenza Pathogenesis
What molecular players and pathways determine disease severity following influenza infection?; this is a question explored by Dr. Lara Mahal's group at the Biomedical Chemistry Institute, Department of Chemistry, New York University, NY. The group specifically wanted to learn the link between host protein glycosylation and disease severity (Heindel et al. 2020).
For these studies, ferrets provided a better model of the human respiratory tract glycans’ distribution than other animal models such as mice. In ferrets infected with the 2009 H1N1 influenza virus, lectin-microarrays enabled Dr. Mahal's group to show that cell surface high-mannose correlates with disease severity and lung injury. Increased high-mannose levels were confirmed in association with damaged lung tissue, as evidenced by consolidation and inflammation. Importantly, increased glycosylation was evident early during influenza infection.
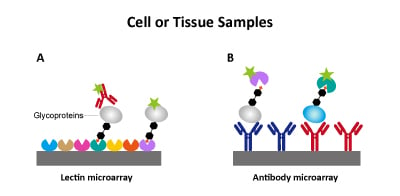
What are lectin microarrays? Lectin-microarrays leverage the specificity of lectins as carbohydrate-binding proteins to detect specific glycan structures. A. Microarrays may use recombinant lectins immobilized onto a solid surface (e.g., glass slide). Detection may be direct by labeling glycoproteins as done by Dr. Mahal's group (Pilobello et al. 2013, Heindel et al. 2020). Alternatively, indirect detection relies on labeled antibodies. B. Immobilized-antibodies may also be used to capture glycoproteins detected by labeled lectins (Hirabayashi et al. 2013). An advantage of lectin-microarrays is analyzing complex cell membranes as those derived from lung tissue samples used by Dr. Mahal's group.
Dr. Mahal's group showed that influenza infection led to increased glycosylation of host proteins, a process that was dependent and downstream to the activation of the unfolded protein response (UPR). To further elucidate the mechanism connecting induced high-mannose to lung damage, the group focused on MBL2, an innate immune lectin known to bind several glycan epitopes, including high-mannose. Because previous studies implicated MBL2 in the severity of influenza infections, Dr. Mahal's group devised in vitro studies that showed the specific reactivity of MBL2 with influenza-infected human cells with high-mannose.
Finally, because the binding of MBL2 is known to activate the complement cascade and consequently induce an inflammatory response, investigators proposed a model whereby the recruitment of MBL2 by high-mannose to influenza-infected lung cells promotes inflammation and tissue damage. Overall, this work identifies new targets that may provide a therapeutic alternative to curve the severe inflammatory responses associated with influenza infections.
Reference
Heindel, D. W. et al. Glycomic analysis of host response reveals high mannose as a key mediator of influenza severity. Proc. Natl. Acad. Sci. U. S. A. (2020) doi:10.1073/pnas.2008203117.
Hirabayashi, J., Yamada, M., Kuno, A. & Tateno, H. Lectin microarrays: Concept, principle and applications. Chem. Soc. Rev. (2013) doi:10.1039/c3cs35419a.
Pilobello, K. T., Agrawal, P., Rouse, R. & Mahal, L. K. Advances in lectin microarray technology: optimized protocols for piezoelectric print conditions. Curr. Protoc. Chem. Biol. (2013) doi:10.1002/9780470559277.ch120035.
- Like (7)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



