Potential Monoclonal Antibody Drugs for Seasonal Influenza
The ongoing SARS-CoV-2 pandemic has boosted the development of new approaches to treat infectious disease. Novel vaccine modalities, such as RNA based formulations developed by Moderna and Pfizer, are starting to emerge as successful approaches based on interim results from Phase 3 trials. Moderna's mRNA vaccine candidate was recently reported to have achieved a ~95% efficacy and shows promise for preventing severe infection.
Another strategy, recombinant monoclonal antibody drugs, as those developed by Regeneron and Eli Lilly, have been recently authorized by the FDA for emergency use. Regeneron's antibody cocktail (casirivimab and imdevimab), authorized for treatment of high-risk patients, has shown clinical success when given early during SARS-CoV-2 infection.
All this recent success makes you wonder when similar strategies will be implemented for other viruses such as influenza viruses, which are associated with high hospitalization burden and mortality rates every year.
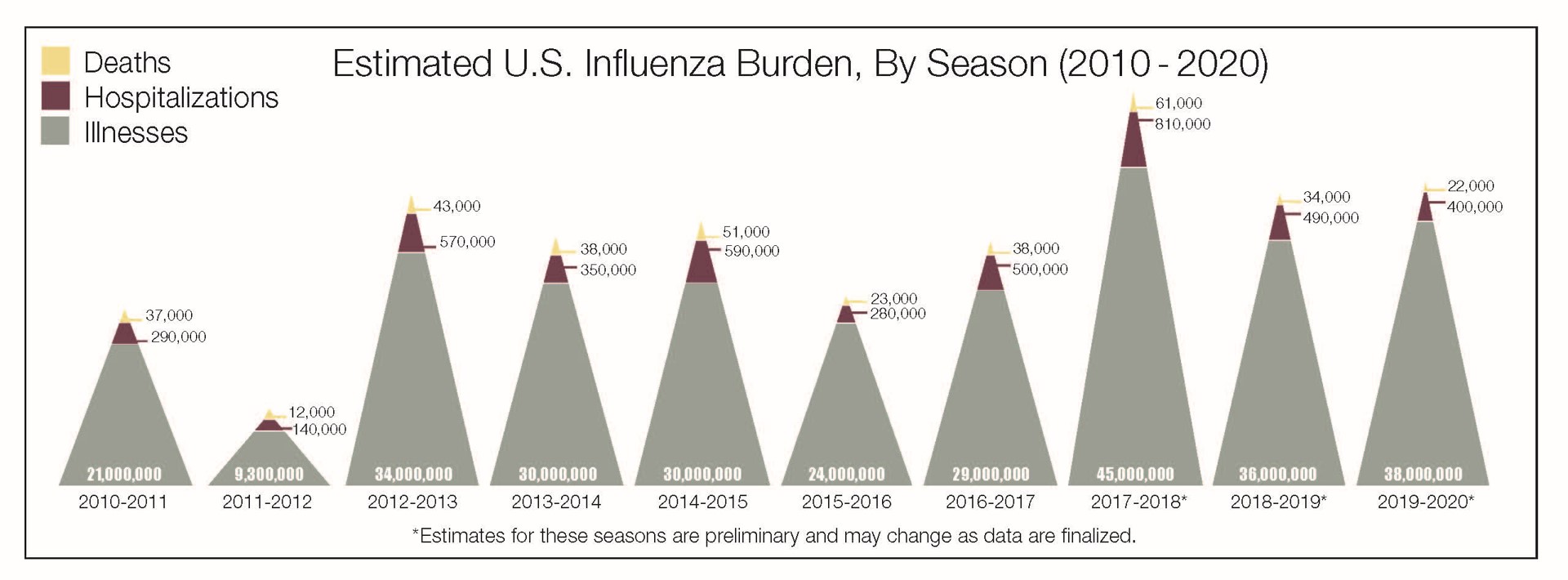
"Source: CDC"; "Materials developed by CDC."
Seasonal Influenza "in a Nutshell"
According to the CDC, seasonal influenza is driven primarily by two types of viruses; influenza type A (subtypes H1N1 and H3N2) and B (linages Yamagata and Victoria). Classification of influenza A viruses under specific subtypes is based on the properties of two surface glycoproteins, hemagglutinin, and neuraminidase. In contrast, influenza B viruses are classified under lineages based on differences in their hemagglutinin glycoprotein.
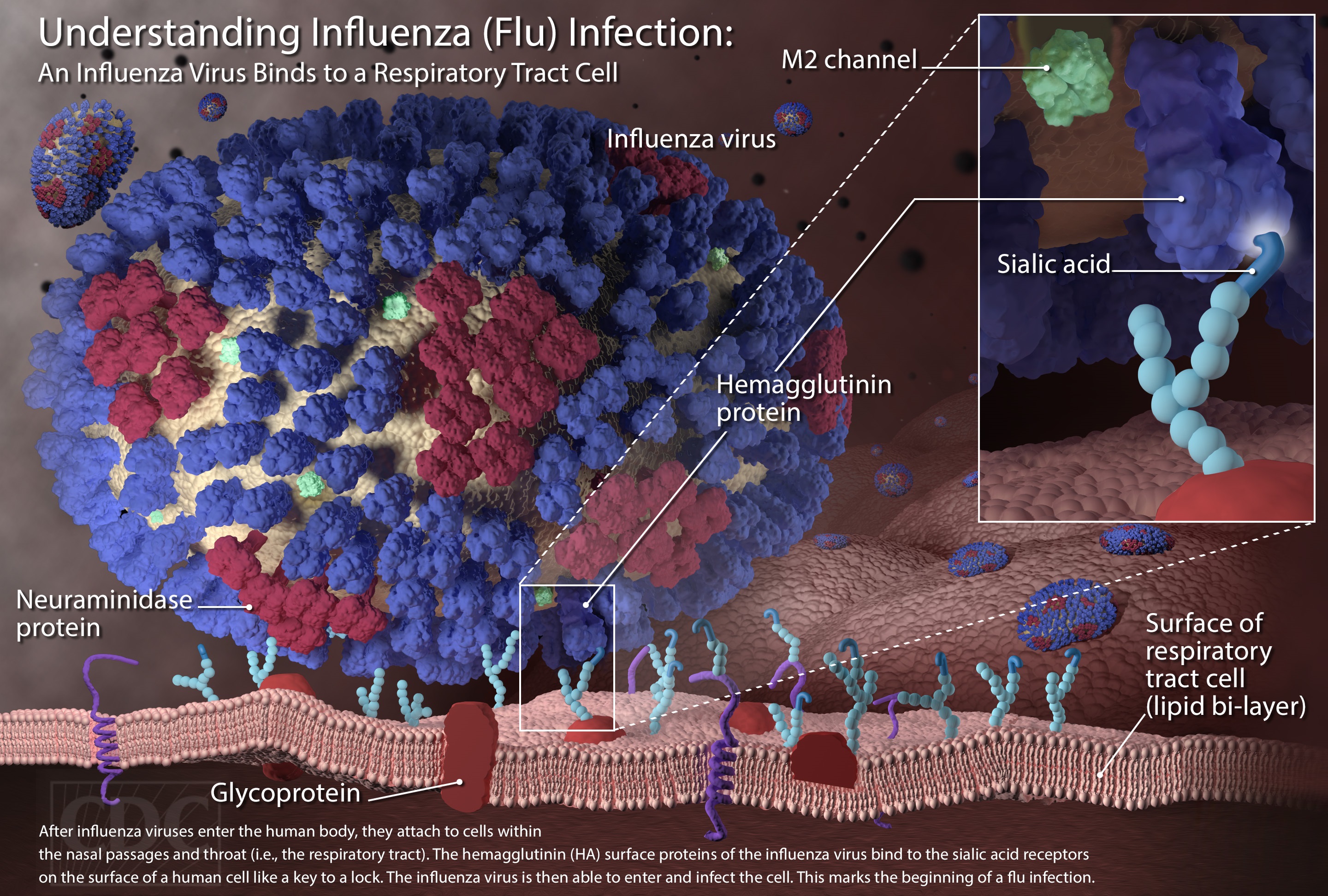
"Source: CDC"; "Materials developed by CDC."
Although influenza A viruses account for the majority of seasonal disease cases, significantly, 25% of influenza cases are due to influenza B viruses, which are known to predominantly affect children (Tan et al. 2018).
Aiming for Universal Therapies Against Influenza
A significant obstacle to long-lasting vaccine coverage for influenza is underscored by the high viral mutational rate, which leads to antigenic drift. Therefore, current management of seasonal influenza relies on yearly immunizations that fail to provide full coverage against the specific circulating strains. Efforts are underway towards developing vaccines and antibodies, which may provide a more absolute protection or "universal coverage."
New strategies aim to expand coverage by re-focusing immunogen selection away from the hemagglutinin's variable region and targeting instead conserved antigens, such as the hemagglutinin's stalk or stem domain and the M2 channel's ectodomain (Nachbagauer et al. 2017). Preclinical and clinical studies are underway to evaluate the immunogenicity, safety, and effectiveness of these approaches (Kostolansky et al. 2020, Pleguezuelos et al. 2020). However, experts believe that recent efforts remain more focused on influenza A viral epitopes and fail to address influenza B coverage (Tan et al. 2018, Madsen et al. 2020).
Identifying Effective Monoclonal Antibodies Targeting Influenza B Virus
Because influenza B virus infections are associated with significant morbidity and mortality, researchers at the University of Bergen, Icahn School of Medicine at Mount Sinai, and Washington University School of Medicine aimed at identifying human monoclonal antibodies specific against influenza B neuraminidase epitopes (Madsen et al. 2020). To achieve this goal, investigators leveraged single-cell sorting to isolate plasmablasts from the peripheral blood of an individual in the acute phase of influenza B viral infection. Screening of expressed antibodies allowed them to identify a total of seven antibodies reactive against neuraminidase, a major surface glycoprotein involved in viral egress. Of these, two monoclonal antibodies (1G05 and 2E01) showed neuraminidase inhibition in a broad range of influenza B virus strains. Additionally, the identified antibodies inhibited viral replication in an in vitro plaque reduction neutralization assay. Collectively, their findings allowed investigators to propose that antibodies specifically targeting the neuraminidase active site, such as 1G05 and 2E01, may also have greater viral neutralizing activity. Preclinical studies where mice were exposed to influenza B strains in circulation demonstrated that both antibodies conferred in vivo protection even following a lethal viral load.
Finally, investigators went on to identify the structural basis for the inhibitory activity of both antibodies, characterizing the features in the antibodies critical for interaction with key conserved residues within the neuraminidase active site pocket. Overall, the identification of these antibodies not only offers a potential therapeutic tool for the treatment of influenza B infections but also may aid in instructing immunogen selection to achieve broader vaccine coverage.
Kostolanský, F., Tomčíková, K., Briestenská, K. & E., M. M. Universal anti-influenza vaccines based on viral HA2 and M2e antigens. Acta Virol. (2020) doi:10.4149/av_2020_408.
Madsen, A. et al. Human Antibodies Targeting Influenza B Virus Neuraminidase Active Site Are Broadly Protective. Immunity (2020) doi:10.1016/j.immuni.2020.08.015.
Nachbagauer, R. & Krammer, F. Universal influenza virus vaccines and therapeutic antibodies. Clinical Microbiology and Infection (2017) doi:10.1016/j.cmi.2017.02.009.
Pleguezuelos, O. et al. Immunogenicity, safety, and efficacy of a standalone universal influenza vaccine, Flu-V, in healthy adults a randomized clinical trial. Ann. Intern. Med. (2020) doi:10.7326/M19-0735.
Tan, J., Asthagiri Arunkumar, G. & Krammer, F. Universal influenza virus vaccines and therapeutics: where do we stand with influenza B virus? Current Opinion in Immunology (2018) doi:10.1016/j.coi.2018.04.002.
- Like (3)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



