New Alzheimer's Biomarkers may Support Early Diagnosis
Alzheimer's disease is a type of neurodegenerative condition most commonly associated with dementia. The disease's progression is slow, and several stages may be identified, including preclinical (preceding symptoms), mild cognitive impairment, and mild to severe dementia. Two main pathological findings are critical to Alzheimer's disease diagnosis, accumulation of extracellular amyloid beta plaques and intracellular phosphorylated tau neurofibrillary tangles. Presently, there are no effective therapies for the disease. Recent Phase 3 clinical trials evaluated the efficacy of Biogen's monoclonal antibody drug, Aducanumab, which targets specifically aggregated beta amyloid. A final FDA decision on the potential approval of Aducanumab is expected in March 2021.
Alzheimer's disease diagnostic strategies currently rely on evaluating the levels of amyloid beta and phosphorylated tau within the cerebrospinal fluid. Specifically, diagnostic immunoassays measure the levels of amyloid beta 42 (Aβ42), total-tau, and phosphorylated tau181 (p-tau181) fragments. The levels of these biomarkers allow clinicians to diagnose or rule out Alzheimer's disease (Leitao et al. 2019). However, only p-tau181 specifically identifies Alzheimer's disease among these biomarkers, and only after mild cognitive impairment is present. Therefore, diagnostic biomarkers able to identify Alzheimer's disease at its earliest preclinical stage are needed.
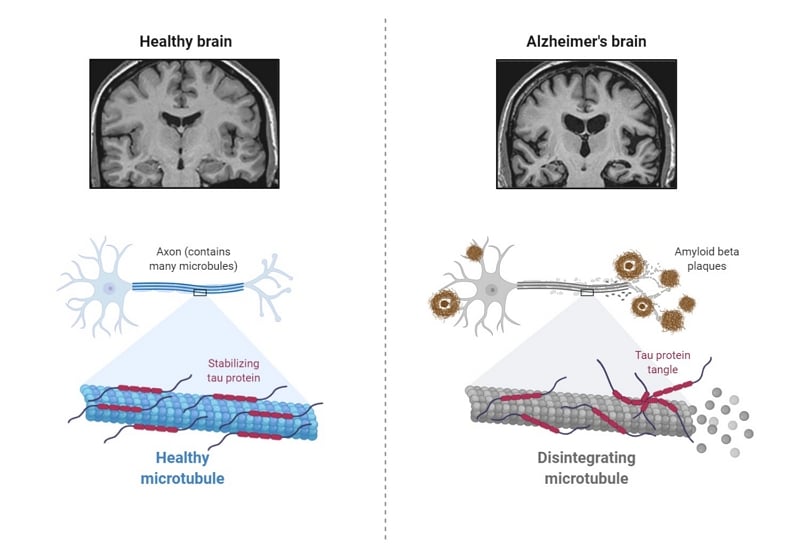
Reprinted from "Pathology of Alzheimer's Disease 2", by BioRender.com (2021). Retrieved from https://app.biorender.com/biorender-templates
Improving Alzheimer's Diagnosis
In a recent study, Karikari et al. 2020 analyzed the levels of two new phosphorylated tau biomarkers, N-p-tau217 and N-p-tau181. To this end, a sandwich ELISA was designed using phosphospecific monoclonal antibodies to capture each p-tau217 and p-tau181, followed by detection with monoclonal antibodies targeting a common N-terminus epitope. This approach differs from current diagnostic immunoassays where p-tau181 fragments are detected by targeting a middle epitope in p-tau181 or M-p-tau181.
Investigators found a better correlation between elevated levels of these two new biomarkers, as opposed to M-p-tau181, and the presence of early mild cognitive impairment. Additionally, both markers were able to identify the earliest sign of disease, amyloid beta pathophysiology, and distinguish between Alzheimer's related mild cognitive impairment and non-Alzheimer's related decline. Compared with the M-p-tau181 conventional marker, these two new biomarkers can diagnose Alzheimer's disease at earlier stages more efficiently.
Similar findings were reported by Suarez-Calvet et al. 2020. Their study also showed that the levels of these two new markers (N-p-tau217 and N-p-tau181) and an additional new biomarker, M-p-tau231, are increased at the early stages of amyloid beta pathology. Importantly, these early tau changes (i.e., N-p-tau181) could also be detected in the plasma, making a less invasive diagnostic approach possible. Overall, these new tau biomarkers' value lies in their improved ability to detect preclinical Alzheimer's. Identifying this early window of opportunity would enable a better understanding of Alzheimer's disease etiology and pathophysiology.
Explore the benefits of Rabbit monoclonals "MonoRab™ Platform" White paper.
Reference
Karikari, T. K. et al. Head-to-head comparison of clinical performance of CSF phospho-tau T181 and T217 biomarkers for Alzheimer’s disease diagnosis. Alzheimer’s Dement. (2020) doi:10.1002/alz.12236.
Leitão, M. J. et al. Clinical validation of the Lumipulse G cerebrospinal fluid assays for routine diagnosis of Alzheimer’s disease. Alzheimer’s Res. Ther. (2019) doi:10.1186/s13195-019-0550-8.
Suarez-Calvet, M. et al. Novel tau biomarkers phosphorylated at T181, T217 or T231 rise in the initial stages of the preclinical Alzheimer’s continuum when only subtle changes in Aβ pathology are detected. EMBO Mol. Med. (2020) doi:10.15252/emmm.202012921.
- Like (1)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



