Antibody-drug Conjugate, a Potential Trojan Horse to Fight Cancer
An Antibody Drug Conjugate (ADC) is a vector-based chemotherapy that relies on the use of a monoclonal antibody (mAb) linked to an anticancer cytotoxic drug (DeCarvalho et al. 1964). Unlike conventional chemotherapy methods, ADCs can selectively target diseased cells, minimizing collateral damage to surrounding healthy tissue and avoiding redundant toxicity. ADCs do this by first binding to target malignant cells through the specificity of antibody-antigen interactions and releasing their cytotoxic payload once internalized by cancer cells, inducing cell death.
For ADCs to work effectively, investigators need to identify suitable antigens that exist exclusively on cancer cells and develop antibodies that bind specifically to these antigens. Theoretically, ADCs can be used to direct effective cytotoxic drugs to kill a broad range of cancer types. However, in practice, numerous obstacles have been encountered during ADC development related to antigen binding that limit broader ADC use.
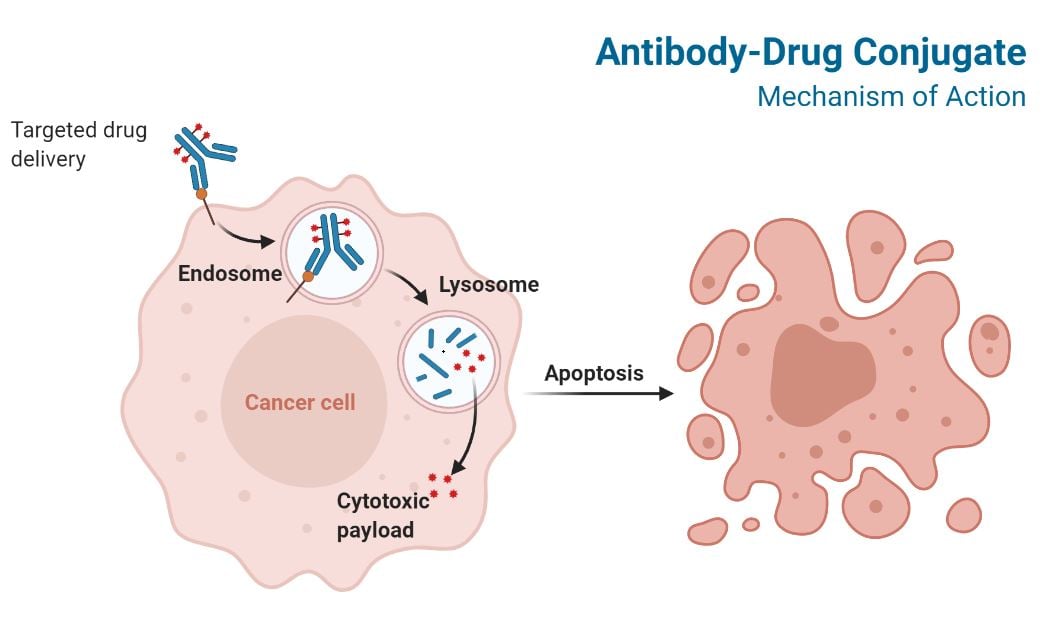
Adapted from “Antibody-Drug Conjugate Mechanism of Action” by BioRender.com (2020). Retrieved from https://app.biorender.com/biorender-templates
Antibody-drug conjugates (ADCs) bind to surface antigens on cancer cells and are internalized through receptor-mediated endocytosis. ADCs are processed through the endosome-lysosome pathway leading to the release of the cytotoxic payload and cancer cell death by inducing DNA damage or affecting microtubule structure (Ponziani et al. 2020).
What are some challenges of ADC-based therapies?
Tumor specific antigens: ADC-based therapies rely on the precise administration of a complex mAb-linker-payload system. For ADC treatment to be successful, the antigens selected as tumor markers must have a relatively high expression level on cancer cells and low or no expression on healthy ones. Moreover, antigens should have an extracellular domain (ECD), which should not be shed into the circulation so that the antibodies bind to targeted cancer cells rather than to circulating ECDs (Nejadmoghaddam et al. 2019).
Antibody specificity: With regard to selecting the ideal antibody for antigen binding, one of the main considerations has been making sure that antibodies can reach the target site. However, mAbs, based on which all current ADCs are constructed, are limited by their large size and do not easily penetrate the tumor microenvironment, especially that of solid tumors (Cruz and Kayser, 2019). Therefore, the limited penetration could compromise ADCs effectiveness.
Creating the ADC: Additionally, it’s also challenging to find an ideal linker with sufficient stability, which is needed to ensure that the ADC can safely circulate through the bloodstream and eventually reach the tumor cells.
The state of ADCs at the Clinic
There are currently nine ADCs approved as cancer treatments by the FDA. Hundreds of clinical trials are ongoing investigating the efficacy of new ADC candidates in treating blood, breast, brain, lung, and other cancers (Drago et al. 2021). However, most of these trials are still in the Phase I stage and it remains to be seen how ADCs will impact cancer treatment.
Despite their growing success, most of the current ADCs in clinical use are manufactured as heterogeneous mixtures, consisting of antibodies linked to a variable number of drugs. This approach presents several challenges for successful ADC therapy, which include competitive inhibition by unloaded antibodies, poor efficacy and increased clearance of ADCs carrying insufficient or too many cytotoxic drugs, respectively. Additionally, because the linkage sites between antibody and -drug payload are not specific, there is a possibility of reduced or altered drug efficacy as a result of the cross-linking process. In order to circumvent this problem, site-specific chemical conjugation of proteins has been investigated for several years. Recently, a research team from the University of Southern California has developed a breakthrough approach for generating site-specific ADCs that ensures the fast release of the drugs into target cells (Dai et al. 2020).
A great deal of effort has also been made by pharmaceutical companies and research institutes to overcome other technological barriers associated with ADC development. As technology advances, we can expect ADCs to evolve with time, eventually improving on the choice of targets, linkers, and cytotoxic payloads.
Reference
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud




Awesome