A Peptide Approach to Reducing alpha-Synuclein in Parkinson's Disease
In Parkinson's disease, the death of substantia nigra dopamine-producing neurons unleashes a sequel of progressive motor and non-motor dysfunctions. There is no cure for this neurodegenerative disease which is only second to Alzheimer's disease in prevalence among the elderly. Parkinson's disease is expected to affect over one million individuals in the US by 2030 (Marras et al. 2018).
The primary pathological mechanism implicated in Parkinson's disease onset is the aggregation of alpha-synuclein; however, other cellular mechanisms at play include abnormal proteostasis and mitochondrial function, as well as increased oxidative stress and inflammation (Jankovic and Tan, 2020). Without a cure in sight, current treatments for Parkinson's disease focus on alleviating symptoms by maintaining or increasing the levels and action of dopamine in the brain. Alternatively, several surgical procedures help reduce symptoms once drugs are ineffective.
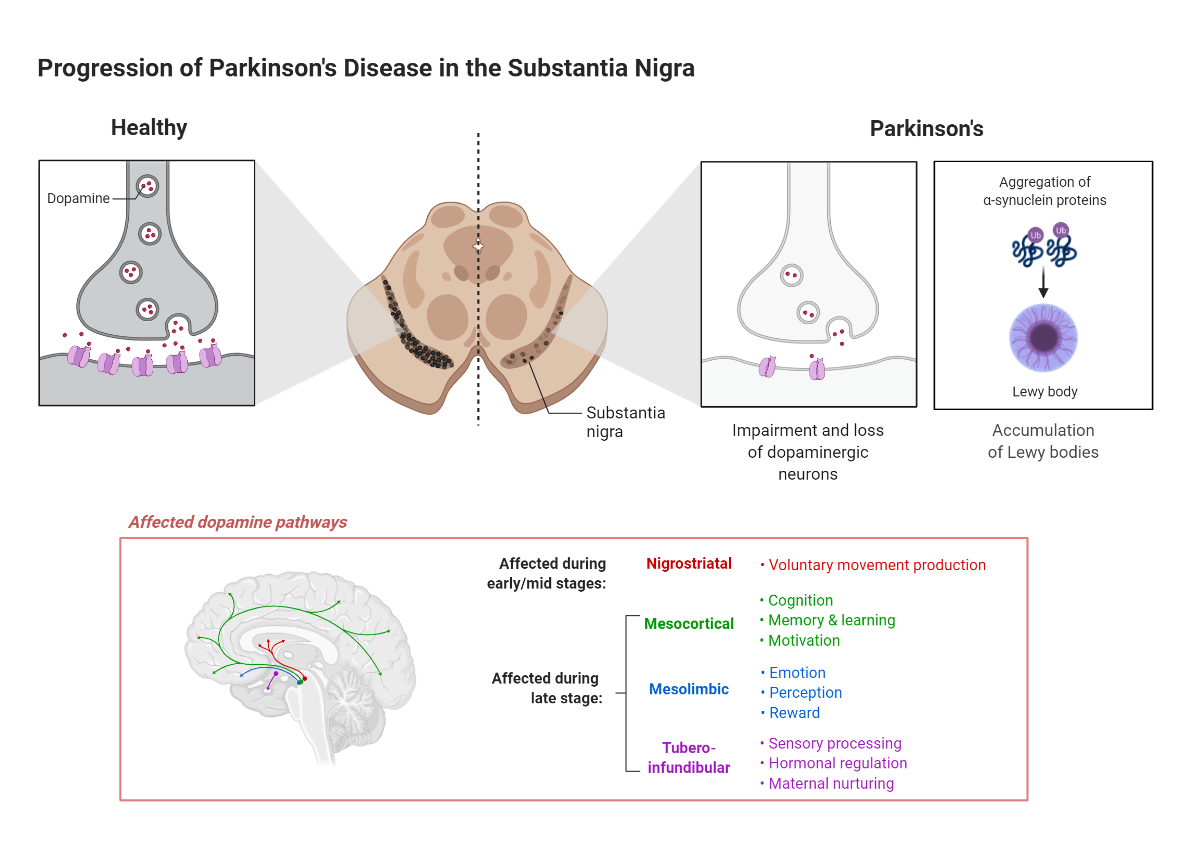
Adapted from "Progression of Parkinson's Disease in the Substantia Nigra” by BioRender.com (2021). Retrieved from https://app.biorender.com/biorender-templates
New therapies are on the horizon for Parkinson's disease, currently at different preclinical to clinical stages. Some strategies aim at slowing disease progression by directly tackling the alpha-synuclein pathology. In contrast, other approaches based on stem cell therapies may provide new alternatives to reduce symptoms.
A Tat-βsyn-degron Peptide Targeting alpha-Synuclein
Misfolding and aggregation of the synaptic protein alpha-synuclein and consequent formation of Lewy bodies are hallmarks of Parkinson's disease (Henderson et al. 2019). Two significant findings strengthen the connection between alpha-synuclein aggregation and disease progression: 1. aggregation of alpha-synuclein in different brain regions correlates with disease progression, and 2.reducing alpha-synuclein expression through knockdown approaches attenuates some motor deficits and neurodegeneration in animal models (Zharikov et al. 2015, Kim et al. 2017). Therefore, strategies specifically targeting alpha-synuclein expression may provide the best opportunities for curving disease progression.
To this end, recent work by Jin and colleagues explored the use of a Tat-βsyn-degron peptide to bind alpha-synuclein and induce its proteasomal degradation specifically. By following this strategy, Jin et al. hoped to improve upon previous RNA-based approaches to knockdown alpha-synuclein expression.
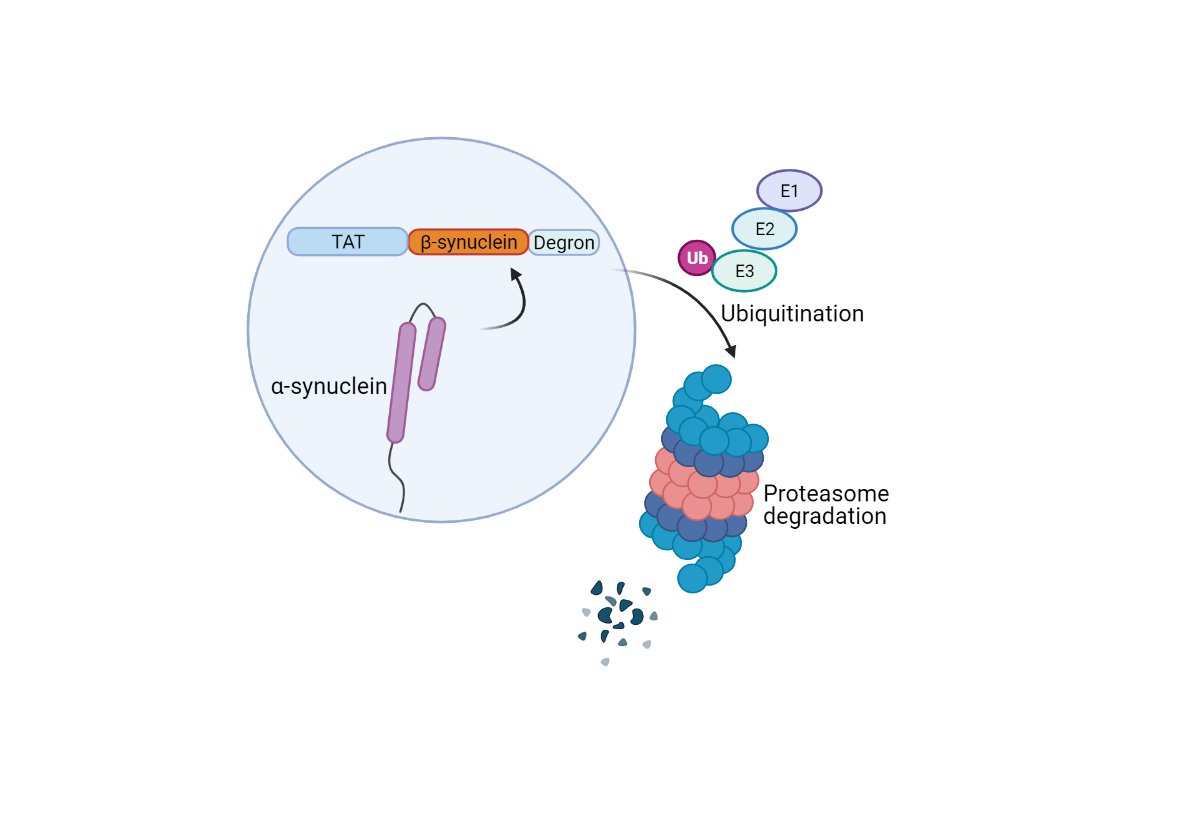
Created with BioRender.com
The Tat-βsyn-degron peptide designed and used by Jin et al. consists of three major domains, a Tat cell-penetrating peptidesequence, an alpha-synuclein binding sequence from beta-synuclein, and a degron motif for proteasome targeting. Jin and colleagues demonstrated specific binding between their Tat-βsyn-degron peptide and alpha-synuclein by using a Biacore platform.
First, Jin and colleagues demonstrated that the Tat-βsyn-degron peptide efficiently reduced the levels of alpha-synuclein in vitro in HEK293 cells and cortical neurons, as well as in vivo in mice overexpressing a mutant human form of alpha-synuclein. The Tat-βsyn-degron peptide effectively reduced dopaminergic neuronal death in an in vitro model of Parkinson's disease. Significantly, by leveraging in vivo models of Parkinson's disease, Jin et al. demonstrated that the peptide reduced the propagation and seeding of alpha-synuclein, decreased dopaminergic neuronal death, and was successful in rescuing motor deficits.
Overall, these findings provide compelling evidence supporting the potential value of directly targeting alpha-synuclein as a therapeutic strategy. Therefore, investigators are hopeful that clinical studies may be undertaken to evaluate this approach's efficacy in slowing down Parkinson's disease progression.
Reference
Henderson, M.X. et al. α-Synuclein pathology in Parkinson's disease and related α-synucleinopathies. Neuroscience letters vol. 709 (2019): 134316. doi:10.1016/j.neulet.2019.134316.
Jankovic, J. & Tan, E. K. Parkinson's disease: Etiopathogenesis and treatment. J. Neurol. Neurosurg. Psychiatry (2020) doi:10.1136/jnnp-2019-322338.
Jin, J.W. et al. Development of an α-synuclein knockdown peptide and evaluation of its efficacy in Parkinson's disease models. Commun Biol. 2021 Feb 19;4(1):232. doi: 10.1038/s42003-021-01746-6.
Kim, Y. C. et al. RNA interference of human α-synuclein in mouse. Front. Neurol. (2017) doi:10.3389/fneur.2017.00013.
Marras, C. et al. Prevalence of Parkinson's disease across North America. npj Park. Dis. (2018) doi:10.1038/s41531-018-0058-0.
Zharikov, A. D. et al. ShRNA targeting α-synuclein prevents neurodegeneration in a Parkinson's disease model. J. Clin. Invest. (2015) doi:10.1172/JCI64502.
- Like (2)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



