Lentiviral-Modified Cells for Rare Disease Therapy: Autologous HPSC therapy for SCID-ADA
Severe combined immunodeficiency (SCID) is a rare group of disorders in which immune responses are compromised by defects in the development and function of T cells. Additionally, in some forms of SCID, B and/or NK cells may also be defective (Fischer, 2000). SCID affects 1/40-75,000 infants and may be brought about by inherited autosomal recessive (e.g., SCID-ADA) or X-linked mutations (i.e., SCID-X1). Mutations in over 20 genes have been linked to SCID, but more remain unidentified (Kumrah et al. 2020). Symptoms of SCID occur early in affected infants, usually presenting as recurrent and severe infections. If left untreated, SCID has a poor prognosis, with infants failing to thrive and succumbing within their first year of life (Haddad and Hoenig, 2019).
Newborn screening panels in the U.S. started to include SCID testing since 2010. Diagnosis relies on the levels of T cell receptor excision circles (TRECs). TRECs are extrachromosomal segments of DNA excised during T cell receptor (TCR) gene rearrangement (Hazenberg et al. 2001). Because TREC numbers correlate with T cell production, newborns with SCID-related mutations have decreased or absent TRECs upon peripheral blood analysis by quantitative-PCR (CDC).
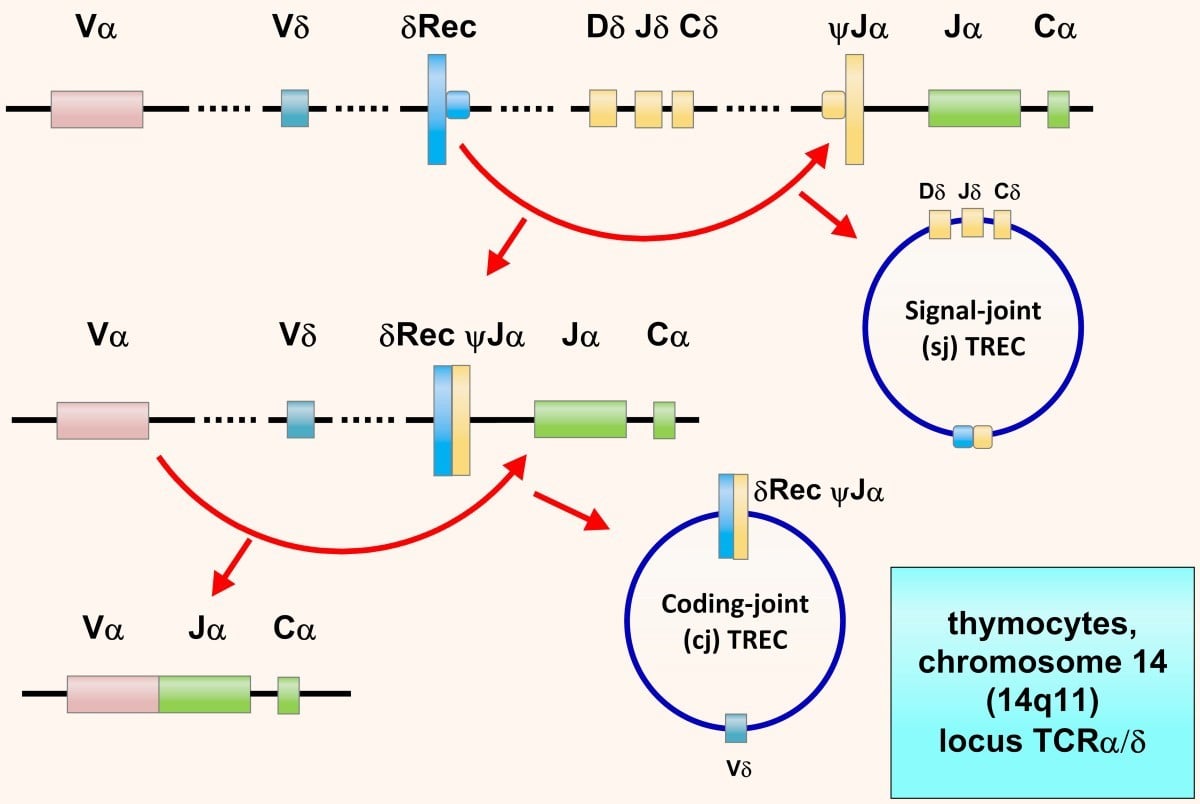
“T cell Receptor Excision Circles (TRECs). TRECs are episomal DNA circles produced in thymocytes by excisional rearrangements of T cell receptor (TCR) genes; they are stable, not duplicated during mitosis, diluted out with each cell division, and therefore higher in thymocytes, recent thymic emigrants (RTEs) and naïve T cells. Quantitative polymerase chain reaction (PCR) of coding-joint (cj) δRec ψJα TREC, produced at TCRα/δ locus within chromosome 14 (14q11) by > 70% of developing human α:β T cells, counts in the peripheral blood naïve α:β T lymphocytes recently dismetted by thymus: in newborn, values < 25 TRECs/μL indicate SCID.” Retrived without modifications from (Cossu, 2019). https://creativecommons.org/licenses/by/2.0/
SCID Therapies
The primary therapeutic approach for SCID relies on hematopoietic stem cell transplantation. Nevertheless, human leukocyte antigen (HLA) compatibility is required for the success of allogeneic hematopoietic stem cell transplantation, excluding a significant portion of affected infants. For one form of the disease, SCID-ADA, involving adenosine deaminase deficiency and consequent lymphocyte toxicity, enzyme replacement therapy (e.g., polyethylene glycol-adenosine deaminase or PEG-ADA) supports removal of the toxic adenosine and deoxyadenosine metabolites and improves immune function. However, ADA replacement therapy is not curative, leading only to partial recovery of immune function. Combined with its high cost, this approach provides a short-term solution (Flinn and Gennery, 2018).
Genetically modified autologous hematopoietic stem cell therapy has shown to be an effective strategy to restore immune function. This approach was leveraged in a clinical trial around a decade ago to restore IL-2 receptor γc gene expression in infants diagnosed with SCID-X1. The trial enrolled 5 infants who received autologous hematopoietic stem cells modified using a replication-defective retrovirus, specifically a Moloney murine leukemia virus vector, to deliver the normal γc gene sequence. Follow-up findings at 10 and 30 months post-treatment confirmed restored T cell function (Hacein-Bey-Abina et al. 2002). Moreover, sustained functional benefits were shown to persist even after 4 years of treatment, as reported by Hacein-Bey-Abina and colleagues in 2003. Nevertheless, an unfortunate outcome for some of the patients was the occurrence of random retroviral integration leading to insertional oncogenesis. Uncontrolled T cell proliferation was reported after ~2.5 years of treatment in two children (Hacein-Bey-Abina et al. 2003).
Lentiviral gene therapy for SCID-ADA
As an alternative and safer approach for gene delivery, new clinical trials have leveraged lentiviral vectors for the ex vivo modification of autologous hematopoietic stem cells intended as therapies for SCID-ADA. Donald B. Kohn and colleagues recently reported the outcome of clinical trials carried on in the U.S. and U.K., which enrolled a total of 50 therapy recipients (Kohn et al. 2021). For these studies, investigators developed a self-inactivating lentiviral vector (EFS-ADA LV).
Findings of follow-up evaluations at 24 months (U.S.) and 36 months (U.K.) post therapy confirmed the treatment’s efficacy, which led to the correction of metabolic toxicity and immunodeficiency in the majority of study participants (48/50). Investigators showed that ADA activity was corrected to normal levels, comparable to detoxifying activities present in healthy children. Additionally, lymphocyte numbers, T and B cells, were corrected to normal levels for most patients allowing termination of immunoglobulin-replacement therapy. Finally, no abnormalities in lymphocyte proliferation occurred in any of the patients, supporting the approach's safety.
Reference
- Like (3)
- Reply
-
Share
About Us · User Accounts and Benefits · Privacy Policy · Management Center · FAQs
© 2026 MolecularCloud



